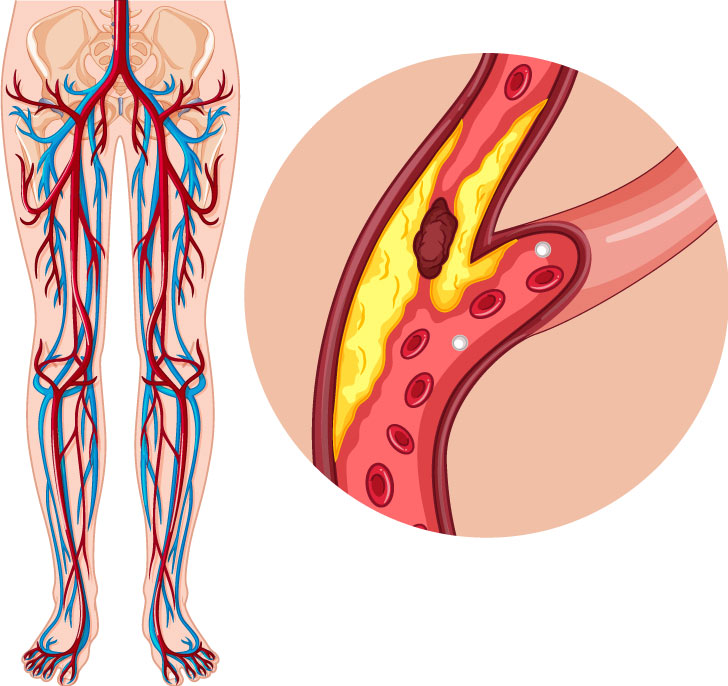
Ra Medical Systems, Inc. announced that the US Food and Drug Administration has granted market clearance for the company’s Dabra (Destruction of Arteriosclerotic Blockages by laser Radiation Ablation) atherectomy system with catheter for the treatment of peripheral artery disease. In October 2016, the company announced that the Dabra system was approved in Europe.
Ehtisham Mahmud, MD, Chair of the pivotal study of the device, explained in the company’s announcement, “Dabra’s pivotal study demonstrated 95% success without any clinically significant adverse events. Furthermore, the Dabra catheter is unique in its ability to cross chronic total occlusions without having to cross the lesion with a wire,and it also debulks and modifies arterial blockages, making it an invaluable clinical tool.” Dr. Mahmud is Chief of Cardiovascular Medicine, Director of Sulpizio Cardiovascular Center-Medicine, and Director of Interventional Cardiology at the UC San Diego School of Medicine in San Diego, California.
On June 1, the company announced the commercial launch of Dabra in the United States. Athar Ansari, MD, with the California Heart & Vascular Clinic in El Centro, California, performed the first postclearance procedure using the Dabra system via brachial artery access to treat a patient with arterial blockages.
Additionally, the company advised that Raghotham Patlola, MD, will present new data on Dabra on June 2 at the New Cardiovascular Horizons Conference in New Orleans, Louisiana. In the presentation,“Novel Wireless Laser for Treating Chronic Total Occlusions,”Dr. Patlola will highlight Dabra’s safety and efficacy, including a 95% success rate with no adverse events in the clinical study. Dr. Patlola is with the Louisiana Cardiovascular & Limb Salvage Center in Lafayette, Louisiana.
Source: https://evtoday.com/news/fda-clears-ra-medicals-dabra-atherectomy-system-to-treat-pad